Volume 6, Issue 2 (Spring 2018- 2018)
PCP 2018, 6(2): 111-118 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Pirkhaefi A. Evaluation of Cognitive Abilities of Different Groups of Sclerosis Patients and Its Comparison With Healthy People. PCP 2018; 6 (2) :111-118
URL: http://jpcp.uswr.ac.ir/article-1-447-en.html
URL: http://jpcp.uswr.ac.ir/article-1-447-en.html
Department of Psychology, Faculty of Psychology and Medical Sciences, Garmsar Branch, Islamic Azad University, Garmsar, Iran. , apirkhaefi@gmail.com
Full-Text [PDF 682 kb]
(2354 Downloads)
| Abstract (HTML) (5609 Views)
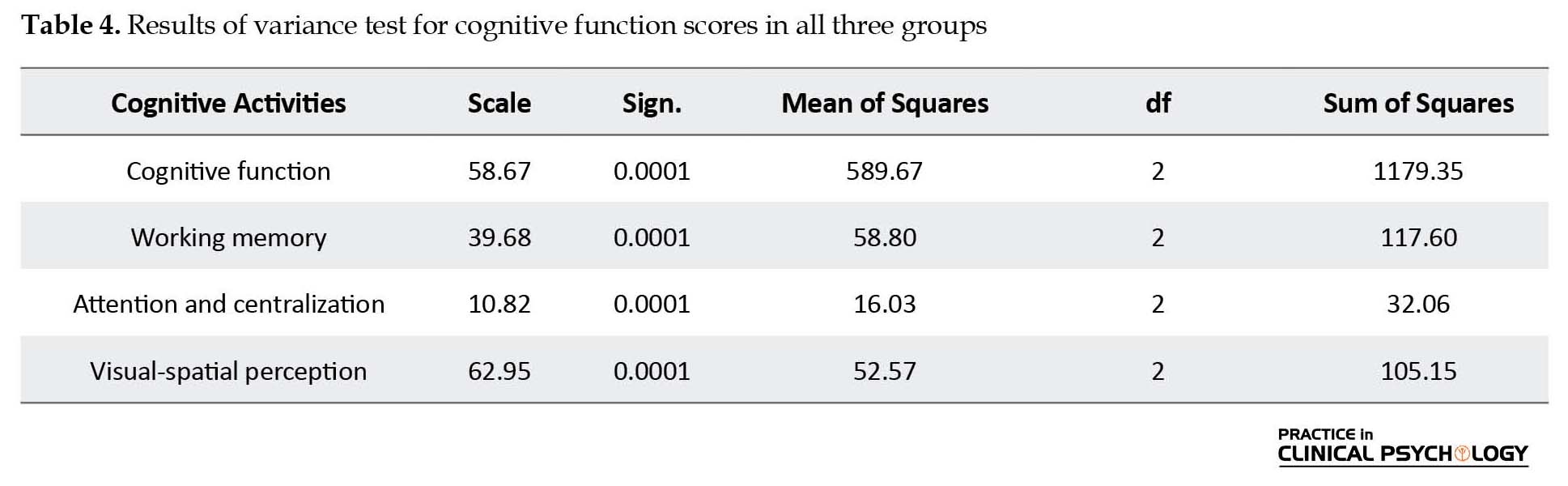
According to the results presented in Table 3, the F ratio among all variables was significant. Table 4 illustrates that cognitive function in various groups showed significant differences with each other. Regarding to the significant differences in variables, an LSD test was used to specify the exact differences among the three groups, and the results are presented in Table 5. As shown in Table 5, significant differences can be seen in the total scores of cognitive function between the three groups of study (RRMS, SPMS, and normal subjects). Additionally, a significant difference was observed among these three groups of subjects in terms of attention, centralization, as well as the visual-spatial perception. In terms of working memory, no significant difference was observed in RRMS and SPMS groups.

4. Discussion
The results of this study showed that there was a significant difference in the overall cognitive status among the three groups (RRMS, SPMS and Normal). Working memory between the two groups (RRMS and SPMS) was not significant, but it was meaningful between RRMS and Normal samples. There were significant differences among the three groups (RRMS, SPMS and Normal) in the attention and centralization function and visual–spatial perception.
Our results showed that the functional cognitive, working memory, attention and centralization, and visual–spatial perception of the RRMS group were better than that of the SPMS group. Although some researchers (like Baddeley, Wilson, 2002) believe in the importance of the MS type in the assessment of cognitive function, especially memory, Whelan-Goodinson et al. (2009) showed that patients with chronic progressive MS in all memory scales had worsened functions in comparison with RRMS patients and that the type of MS has low prediction power of memory function. Other investigators (Rao, Leo, Bernardin, & Unverzagt, 1991; Thornton, & Raz, 1997; Reio, Czarnolewski, & Eliot, 2004; Feinstein, et al., 2013; Bergendal, 2014) revealed that there was a reduction in memory function in cerebrospinal MS patients, indicating that damage in memory of MS patients can represent a moderate subcortical level.
Posner and Rothbart (1998) and Langdon (2011) determined anterior and posterior attention parts (attention section) on forehead and parietal parts, respectively. The anterior and posterior parts support attention within the various assignments, which indicate that these sections may play a role in the regulation of activation in the sections related to the brain cortex. Thus, this suggests increased damage to these brain regions, especially frontal lobe lesions in SPMS patients, where atrophy in attention and centralization ability is more pronounced. Studies (Warren, 2010; Das Nair, et al., 2016; Guimarães, & sa, 2012) have showed that lower scores in visual-spatial perception of SPMS patients represent the effect of right hemisphere damage on visual spatial ability. This finding is in coordination with the findings of other studies (Rourke, & Convay, 1997; Spring, & Deutsch, 1998; Harnadek, & Rourke, 1994; Fuerst, 1990; Amato, et al., 2010; Overell, 2011; Bergendal, 2014; Langdon, 2011).
A weakness in visual-spatial perception in patients with SPMS indicates increased atrophy in the right hemisphere of the brain, which has a greater effect on visual-spatial perception. Thus, these patients have a lower performance in visual-spatial perception than RRMS patients and normal persons. This finding is in coordination with the findings of other researchers (Sawcer, et al., 2014; Schoonheim, et al., 2015; Feinstein, et al., 2013).
Based on the results of this study, which is consistent with other studies, it can be concluded that SPMS group have larger problems than the RRMS group in most cognitive functions. RRMS group also has a bigger problem than the Normal group. In fact, the most important differences were found in the work memory and visual–spatial perception tasks between RRMS and SPMS groups with the normal group.
The most important limitation of this research was the inability of the researchers to use more precise neuropsychological tasks or tools. In addition, it was difficult to completely match the sample. Thus, it is suggested that future studies should include more controlled research designs and more precise tools for comparing MS patients.
The results showed that there is a difference between cognitive abilities of patients and healthy people. Interestingly, there was a significant difference among the different groups of sclerosis. In this study, the SPMS group was weaker in cognitive abilities compared to the RRMS group. The results of this study can be generalized in the range of patients examined, and it is suggested to be examined with other samples.
Acknowledgments
We are thankful to the community supporting MS patients for the cooperation and support. We are also grateful to the patients who have collaborated with us. This study was conducted with the financial support of psychology departments of Islamic Azad University of Garmsar and Semnan branch.
Conflict of Interest
The authors declared no conflicts of interest.
References
Full-Text: (1711 Views)
1. Introduction
Multiple Sclerosis (MS) is a neurodegenerative disease in which the immunity system acts against the non-neuronal cells of the brain that produce myelin (Compston, & Coles, 2002). Its exact cause has not been identified yet, but it seems that an infectious disease with a slow progression causes such a disorder whose clinical symptoms might be observed at a young age (Kantarci, & Wingerchukb, 2006). MS can be categorized into recurrent-remittent, progressive-relapsing, primary progressive, and secondary progressive types. Globally, 2.5 million people suffer from MS; statistics show that 250000 to 350000 individuals have MS in the United States (Guimarães, & sa, 2012; Mohammadi Rezve, & Afshar, 2009). The US MS society declared the number of MS patients to be around 30000 in 2003, and currently, there are more than 40000 people who suffer from MS in the country, with about 2000 new patients being added annually to this population. Unfortunately, the number of MS patients in the US is higher than that of the neighboring countries while its prevalence is unknown. In Iran, the prevalence rate is between 15 and 30 per 100000 people (Hasanzadeh, Fallahi Khoshknab, & Norozi, 2012).
Studies have shown MS involves the physical body, and 50-60% of MS patients present with cognitive impairments. Thus, cognitive impairment could be considered as one of the major symptoms of this disease observed through all clinical stages of the illness (Amato, et al., 2010; Langdon, 2011; Yektamaram, & Nazmdeh, 2009). Cognitive functions refer to some inner process of the mind such as attention, consciousness, problem solving, perception, recognition, language, and decision. In fact, some of the functions like organization, making decision, working memory, attention and concentration, motion control, feeling and understanding time, future prediction, regeneration, inner language, and problem solving are the most important performances of the brain that are helpful in life, learning, and intelligent responses of individuals (Welsh, & Pennington, 1988; Feuillet, et al., 2007).
Working memory is an executive system that consists of memory components in different parts of the brain and uses data representation for different executive functions (Warren, 2010). To evaluate working memory in patients with MS, research works have focused on the phonological loop that causes cognitive impairment in these patients. Different levels of paramnesia in MS patients are related to some issues like the length of illness and its severity, having mood disorders, and types of MS (Das Nair, Martin, & Lincoln, 2012). The results of some studies were representative of the weakness of semantic memory in these patients (Goldstein, McKendall, & Haut, 1992; Overell, 2011). However, investigations showed that MS patients having severe depression suffered from significant injuries in working memory, procedural memory, and declarative memory (Grigsby, & Foonge, 1993; Chelune, Feisthamel, & Stone, 2004; Sawcer, Franklin, & Ban, 2014).
Attention and centralization are the other functions affected by the MS disease. Centralization is a general attempt and an increased state of attention to overlook the non-related stimuli. To assess the relationship between this kind of amnesia and the type of disease, researchers have evaluated memory functions in MS patients. Although Beatty and Monson (1990) showed that patients with SPMS have weak functions in all memory aspects compared to those with RRMS, researchers have stated that the type of MS disease has a low ability to predict the memory function (Beatty, & Monson, 1990; Ling, & Selby, 1998; Schoonheim, et al., 2015). Moreover, researchers have also assessed the correlation between reducing the memory of capacity with place of plaques in brain has been assessed.
It has been reported that the intelligent function and memory of MC patients, based on the disease type (cerebral, cerebo-spinal, or spinal) and MRI findings, are not the same, whereas the scores of verbal memory in cerebral or cerebo-spinal patients were lower than in spinal patients. Studies have also showed that memory injuries in MS patients could be a sign of dementia (Cohen, 1999; Filippi, et al., 1994; Feinstein, DeLuca, Baune, Filippi, & Lassman 2013; Bergendal 2014). Cognitive changes in MS patients could affect their daily life and even interfere with their social and occupational action (Bassett, 2005). Unfortunately, there are limited studies regarding cognitive functions of different groups of MS sufferers in Iran. Most of the studies conducted in Iran are in the field of genetics and therapeutics, and less attention has been paid to the cognitive aspects of the disease. In addition, there are less cognitive differences in the types of MS in Iran. For this reason, the present study was designed and conducted with the aim of evaluating the cognitive abilities of different groups of sclerosis patients and comparing them with those of healthy people.
2. Methods
This study has been done by post-event method. The statistical population included all adults with MS diagnosis who were referred to the community supporting MS patients in Tehran during the year 2012. The age range of these patients was 20 to 55 years. In the community, the diagnosis of the patients was confirmed by a specialist physician. The present study sample consisted of 30 RRMS patients, 30 SPMS patients, and 30 normal persons (39 males and 51 females). They were selected using the purposive sampling method. Inclusion criteria were age (20-55 years), diagnosis of MS at least six months prior to the investigation, capability in understanding the Persian language, and having tendency and informed consent to participate in the research, referral to an MS specialized physician and having a medical file there.
The Montreal Cognitive Assessment Scale (MCA)was proposed by Ziad Nasreddine (1996) in Montreal (Nasreddine, et al., 2005). The scale contains 6 subtests. The highest scores in visual-spatial perception, attention and centralization, working memory, and language is 5; in the executive function is 4; and in orientation is 6. The test time lasts from 10 to 15 minutes (Gill, Freshman, Blender, & Ravina, 2008). The Cronbach’s alpha reliability of this scale has been reported to be 0.77, and the correlation between those scores with a brief examination and mental status scores has been reported to be 0.79 at a significance level of 0.05. This assessment can diagnose 85.7% of healthy adults and 90% of individuals with cognitive impairments accurately (Amsaki, Molavi, Chitsaz, Movahedehabtahi, Asgarii, 2011). In this study, Cronbach’s alpha was 0.78.
The research was carried out in the community supporting MS patients located on Haft Tir Avenue in Tehran. The test data were collected by the researcher and his assistant during the 2 months of the summer. The test was performed individually and took 10 15 minutes for each patient. Tailored consent and confidentiality agreement of information were obtained from the research sample.
3. Results
The results of the descriptive findings are given in Tables 1 and 2, which shows the mean and standard deviation (SD) of cognitive functions scores in three groups (RRMS, SPMS, and healthy persons). The Multivariate Analysis of Variance test (MANOVA) was used to show the difference between the groups. The MANOVA findings are presented in Tables. The F ratios achieved by the MANOVA test are significant in 0.0001 level; therefore, the main research hypothesis showed the difference between the groups.
Multiple Sclerosis (MS) is a neurodegenerative disease in which the immunity system acts against the non-neuronal cells of the brain that produce myelin (Compston, & Coles, 2002). Its exact cause has not been identified yet, but it seems that an infectious disease with a slow progression causes such a disorder whose clinical symptoms might be observed at a young age (Kantarci, & Wingerchukb, 2006). MS can be categorized into recurrent-remittent, progressive-relapsing, primary progressive, and secondary progressive types. Globally, 2.5 million people suffer from MS; statistics show that 250000 to 350000 individuals have MS in the United States (Guimarães, & sa, 2012; Mohammadi Rezve, & Afshar, 2009). The US MS society declared the number of MS patients to be around 30000 in 2003, and currently, there are more than 40000 people who suffer from MS in the country, with about 2000 new patients being added annually to this population. Unfortunately, the number of MS patients in the US is higher than that of the neighboring countries while its prevalence is unknown. In Iran, the prevalence rate is between 15 and 30 per 100000 people (Hasanzadeh, Fallahi Khoshknab, & Norozi, 2012).
Studies have shown MS involves the physical body, and 50-60% of MS patients present with cognitive impairments. Thus, cognitive impairment could be considered as one of the major symptoms of this disease observed through all clinical stages of the illness (Amato, et al., 2010; Langdon, 2011; Yektamaram, & Nazmdeh, 2009). Cognitive functions refer to some inner process of the mind such as attention, consciousness, problem solving, perception, recognition, language, and decision. In fact, some of the functions like organization, making decision, working memory, attention and concentration, motion control, feeling and understanding time, future prediction, regeneration, inner language, and problem solving are the most important performances of the brain that are helpful in life, learning, and intelligent responses of individuals (Welsh, & Pennington, 1988; Feuillet, et al., 2007).
Working memory is an executive system that consists of memory components in different parts of the brain and uses data representation for different executive functions (Warren, 2010). To evaluate working memory in patients with MS, research works have focused on the phonological loop that causes cognitive impairment in these patients. Different levels of paramnesia in MS patients are related to some issues like the length of illness and its severity, having mood disorders, and types of MS (Das Nair, Martin, & Lincoln, 2012). The results of some studies were representative of the weakness of semantic memory in these patients (Goldstein, McKendall, & Haut, 1992; Overell, 2011). However, investigations showed that MS patients having severe depression suffered from significant injuries in working memory, procedural memory, and declarative memory (Grigsby, & Foonge, 1993; Chelune, Feisthamel, & Stone, 2004; Sawcer, Franklin, & Ban, 2014).
Attention and centralization are the other functions affected by the MS disease. Centralization is a general attempt and an increased state of attention to overlook the non-related stimuli. To assess the relationship between this kind of amnesia and the type of disease, researchers have evaluated memory functions in MS patients. Although Beatty and Monson (1990) showed that patients with SPMS have weak functions in all memory aspects compared to those with RRMS, researchers have stated that the type of MS disease has a low ability to predict the memory function (Beatty, & Monson, 1990; Ling, & Selby, 1998; Schoonheim, et al., 2015). Moreover, researchers have also assessed the correlation between reducing the memory of capacity with place of plaques in brain has been assessed.
It has been reported that the intelligent function and memory of MC patients, based on the disease type (cerebral, cerebo-spinal, or spinal) and MRI findings, are not the same, whereas the scores of verbal memory in cerebral or cerebo-spinal patients were lower than in spinal patients. Studies have also showed that memory injuries in MS patients could be a sign of dementia (Cohen, 1999; Filippi, et al., 1994; Feinstein, DeLuca, Baune, Filippi, & Lassman 2013; Bergendal 2014). Cognitive changes in MS patients could affect their daily life and even interfere with their social and occupational action (Bassett, 2005). Unfortunately, there are limited studies regarding cognitive functions of different groups of MS sufferers in Iran. Most of the studies conducted in Iran are in the field of genetics and therapeutics, and less attention has been paid to the cognitive aspects of the disease. In addition, there are less cognitive differences in the types of MS in Iran. For this reason, the present study was designed and conducted with the aim of evaluating the cognitive abilities of different groups of sclerosis patients and comparing them with those of healthy people.
2. Methods
This study has been done by post-event method. The statistical population included all adults with MS diagnosis who were referred to the community supporting MS patients in Tehran during the year 2012. The age range of these patients was 20 to 55 years. In the community, the diagnosis of the patients was confirmed by a specialist physician. The present study sample consisted of 30 RRMS patients, 30 SPMS patients, and 30 normal persons (39 males and 51 females). They were selected using the purposive sampling method. Inclusion criteria were age (20-55 years), diagnosis of MS at least six months prior to the investigation, capability in understanding the Persian language, and having tendency and informed consent to participate in the research, referral to an MS specialized physician and having a medical file there.
The Montreal Cognitive Assessment Scale (MCA)was proposed by Ziad Nasreddine (1996) in Montreal (Nasreddine, et al., 2005). The scale contains 6 subtests. The highest scores in visual-spatial perception, attention and centralization, working memory, and language is 5; in the executive function is 4; and in orientation is 6. The test time lasts from 10 to 15 minutes (Gill, Freshman, Blender, & Ravina, 2008). The Cronbach’s alpha reliability of this scale has been reported to be 0.77, and the correlation between those scores with a brief examination and mental status scores has been reported to be 0.79 at a significance level of 0.05. This assessment can diagnose 85.7% of healthy adults and 90% of individuals with cognitive impairments accurately (Amsaki, Molavi, Chitsaz, Movahedehabtahi, Asgarii, 2011). In this study, Cronbach’s alpha was 0.78.
The research was carried out in the community supporting MS patients located on Haft Tir Avenue in Tehran. The test data were collected by the researcher and his assistant during the 2 months of the summer. The test was performed individually and took 10 15 minutes for each patient. Tailored consent and confidentiality agreement of information were obtained from the research sample.
3. Results
The results of the descriptive findings are given in Tables 1 and 2, which shows the mean and standard deviation (SD) of cognitive functions scores in three groups (RRMS, SPMS, and healthy persons). The Multivariate Analysis of Variance test (MANOVA) was used to show the difference between the groups. The MANOVA findings are presented in Tables. The F ratios achieved by the MANOVA test are significant in 0.0001 level; therefore, the main research hypothesis showed the difference between the groups.
According to the results presented in Table 3, the F ratio among all variables was significant. Table 4 illustrates that cognitive function in various groups showed significant differences with each other. Regarding to the significant differences in variables, an LSD test was used to specify the exact differences among the three groups, and the results are presented in Table 5. As shown in Table 5, significant differences can be seen in the total scores of cognitive function between the three groups of study (RRMS, SPMS, and normal subjects). Additionally, a significant difference was observed among these three groups of subjects in terms of attention, centralization, as well as the visual-spatial perception. In terms of working memory, no significant difference was observed in RRMS and SPMS groups.

4. Discussion
The results of this study showed that there was a significant difference in the overall cognitive status among the three groups (RRMS, SPMS and Normal). Working memory between the two groups (RRMS and SPMS) was not significant, but it was meaningful between RRMS and Normal samples. There were significant differences among the three groups (RRMS, SPMS and Normal) in the attention and centralization function and visual–spatial perception.
Our results showed that the functional cognitive, working memory, attention and centralization, and visual–spatial perception of the RRMS group were better than that of the SPMS group. Although some researchers (like Baddeley, Wilson, 2002) believe in the importance of the MS type in the assessment of cognitive function, especially memory, Whelan-Goodinson et al. (2009) showed that patients with chronic progressive MS in all memory scales had worsened functions in comparison with RRMS patients and that the type of MS has low prediction power of memory function. Other investigators (Rao, Leo, Bernardin, & Unverzagt, 1991; Thornton, & Raz, 1997; Reio, Czarnolewski, & Eliot, 2004; Feinstein, et al., 2013; Bergendal, 2014) revealed that there was a reduction in memory function in cerebrospinal MS patients, indicating that damage in memory of MS patients can represent a moderate subcortical level.
Posner and Rothbart (1998) and Langdon (2011) determined anterior and posterior attention parts (attention section) on forehead and parietal parts, respectively. The anterior and posterior parts support attention within the various assignments, which indicate that these sections may play a role in the regulation of activation in the sections related to the brain cortex. Thus, this suggests increased damage to these brain regions, especially frontal lobe lesions in SPMS patients, where atrophy in attention and centralization ability is more pronounced. Studies (Warren, 2010; Das Nair, et al., 2016; Guimarães, & sa, 2012) have showed that lower scores in visual-spatial perception of SPMS patients represent the effect of right hemisphere damage on visual spatial ability. This finding is in coordination with the findings of other studies (Rourke, & Convay, 1997; Spring, & Deutsch, 1998; Harnadek, & Rourke, 1994; Fuerst, 1990; Amato, et al., 2010; Overell, 2011; Bergendal, 2014; Langdon, 2011).
A weakness in visual-spatial perception in patients with SPMS indicates increased atrophy in the right hemisphere of the brain, which has a greater effect on visual-spatial perception. Thus, these patients have a lower performance in visual-spatial perception than RRMS patients and normal persons. This finding is in coordination with the findings of other researchers (Sawcer, et al., 2014; Schoonheim, et al., 2015; Feinstein, et al., 2013).
Based on the results of this study, which is consistent with other studies, it can be concluded that SPMS group have larger problems than the RRMS group in most cognitive functions. RRMS group also has a bigger problem than the Normal group. In fact, the most important differences were found in the work memory and visual–spatial perception tasks between RRMS and SPMS groups with the normal group.
The most important limitation of this research was the inability of the researchers to use more precise neuropsychological tasks or tools. In addition, it was difficult to completely match the sample. Thus, it is suggested that future studies should include more controlled research designs and more precise tools for comparing MS patients.
The results showed that there is a difference between cognitive abilities of patients and healthy people. Interestingly, there was a significant difference among the different groups of sclerosis. In this study, the SPMS group was weaker in cognitive abilities compared to the RRMS group. The results of this study can be generalized in the range of patients examined, and it is suggested to be examined with other samples.
Acknowledgments
We are thankful to the community supporting MS patients for the cooperation and support. We are also grateful to the patients who have collaborated with us. This study was conducted with the financial support of psychology departments of Islamic Azad University of Garmsar and Semnan branch.
Conflict of Interest
The authors declared no conflicts of interest.
References
- Amato, M. P., Portaccio, E., Goretti, B., Zipoli, V., Iudice, A., Pina, D. D. (2010). Relevance of cognitive deterioration in early relapsing-remitting MS: A 3-year follow-up study. Multiple Sclerosis Journal, 16(12), 1474–82. doi: 10.1177/1352458510380089
- Amsaki, G., Molavi, H., Chitsaz, A., Movahedehabtahi, M., asgarii, K. (2011). [Psychometric properties of the Montreal Cognitive Assessment (MoCA) in Parkinson's disease patients in Isfahan (Persian]. Journal of Isfahan Medical School, 29(158), 1606-15.
- Baddeley, A., & Wilson, B. A. (2002). Prose recall and amnesia: Implications for the structure of working memory. Neuropsychologia, 40(10), 1737–43. doi: 10.1016/s0028-3932(01)00146-4
- Bassett, SS. (2005). Cognitive impairment in Parkinson’s disease. Primary Psychiatry, 12(7), 50-5.
- Beatty, W. W., & Monson, N. (1990). Semantic priming in multiple sclerosis. Bulletin of the Psychonomic Society, 28(5), 397–400. doi: 10.3758/bf03334049
- Bergendal, G. (2014). Cognition in multiple sclerosis with special emphasis on MRI finding and cerebrosterol. Solna: Karolinska Institutet.
- Chelune, G. J., Feisthamel, K., & Stone, L. (2004). Assessing the prevalence and relative risk of cognitive dysfunction in patients with multiple sclerosis. Brain Impair, 5, 77.
- Cohen, H. S. (1999). Neuroscience for rehabilitation. Philadelphia: Lippincott Williams & Wilkins.
- Compston, A. Coles, A. (2002). Multiple sclerosis. Lancet, 359, 1221-31.
- Das Nair, R., Martin, K. J., & Lincoln, N. B. (2016). Memory rehabilitation for people with multiple sclerosis. Cochrane Database of Systematic Reviews. 3, CD008754. doi: 10.1002/14651858.cd008754.pub3
- Feinstein, A. (2004). The neuropsychiatry of multiple sclerosis. The Canadian Journal of Psychiatry, 49(3), 157–63. doi: 10.1177/070674370404900302
- Feinstein, A., DeLuca, J., Baune, B. T., Filippi, M., & Lassman, H. (2013). Cognitive and neuropsychiatric disease manifestations in MS. Multiple Sclerosis and Related Disorders, 2(1), 4–12. doi: 10.1016/j.msard.2012.08.001
- Feuillet, L., Reuter, F., Audoin, B., Malikova, I., Barrau, K., Cherif, A. A., et al. (2007). Early cognitive impairment in patients with clinically isolated syndrome suggestive of multiple sclerosis. Multiple Sclerosis Journal, 13(1), 124–7. doi: 10.1177/1352458506071196
- Filippi, M., Alberoni, M., Martinelli, V., Sirabian, G., Bressi, S., Canal, N., et al. (1994). Influence of clinical variables on neuropsychological performance in multiple sclerosis. European Neurology, 34(6), 324–8. doi: 10.1159/000117071
- Foong, J., Rozewicz, L., Quaghebeur, G., Thompson, A. J., Miller, D. H., & Ron, M. A. (1998). Neuropsychological deficits in multiple sclerosis after acute relapse. Journal of Neurology, Neurosurgery & Psychiatry, 64(4), 529–32. doi: 10.1136/jnnp.64.4.529
- Fuerst, D. R., Fisk, J. L., & Rourke, B. P. (1990). Psychosocial functioning of learning-disabled children: Relations between WISC Verbal IQ Performance IQ discrepancies and personality subtypes. Journal of Consulting and Clinical Psychology, 58(5), 657–60. doi: 10.1037/0022-006x.58.5.657
- Gill, D. J., Freshman, A., Blender, J. A., & Ravina, B. (2008). The Montreal cognitive assessment as a screening tool for cognitive impairment in Parkinson's disease. Movement Disorders, 23(7), 1043-6. doi: 10.1002/mds.22017
- Goldstein, F. C., McKendall, R. R., & Haut, M. W. (1992). Gist recall in multiple sclerosis. Archives of Neurology, 49(10), 1060–4. doi: 10.1001/archneur.1992.00530340080021
- Grigsby, J., Ayarbe, S. D., Kravcisin, N., & Busenbark, D. (1993). Working memory impairment among persons with chronic progressive multiple sclerosis. Journal of Neurology, 241(3), 125–31. doi: 10.1007/bf00868338
- Guimarães, J., & Sá, M. J. (2012). Cognitive dysfunction in multiple sclerosis. Frontiers in Neurology, 3, 47. doi: 10.3389/fneur.2012.00074
- Harnadek, M. C. S., & Rourke, B. P. (1994). Principal identifying features of the syndrome of nonverbal learning disabilities in children. Journal of Learning Disabilities, 27(3), 144–54. doi: 10.1177/002221949402700303
- Hasanzadeh, P., Fallahi Khoshknab, M., & Norozi, K. (2012). [Impacts of journaling on anxiety and stress in multiple sclerosis patients (Persian)]. Complementary Medicine Journal of faculty of Nursing & Midwifery, 2(2), 183-93.
- Kantarci, O., & Wingerchuk, D. (2006). Epidemiology and natural history of multiple sclerosis: new insights. Current Opinion in Neurology, 19(3), 248–54. doi: 10.1097/01.wco.0000227033.47458.82
- Langdon, D. W. (2011). Cognition in multiple sclerosis. Current Opinion in Neurology, 24(3), 244–9. doi: 10.1097/wco.0b013e328346a43b
- Ling, N. D., & Selby, M. J. (1998). Assessment of memory in multiple sclerosis patients using the memory assessment scale. Perceptual and Motor Skills, 86(3), 987–98. doi: 10.2466/pms.1998.86.3.987
- Mohammadi Rezve, N., Afshar, H. (2009). [Determining psychological and social factors causing stress in patients with multiple sclerosis (Persian)]. Journal of Fundamentals of Mental Health, 10(40), 305-10.
- Nasreddine, Z. S., Phillips, N. A., Bédirian, V., Charbonneau, S., Whitehead, V., Collin, I., et al. (2005). The Montreal cognitive assessment, MoCA: A brief screening tool for mild cognitive impairment. Journal of the American Geriatrics Society, 53(4), 695–9. doi: 10.1111/j.1532-5415.2005.53221.x
- Posner, M. I., & Rothbart, M. K. (1998). Attention, self-regulation and consciousness. Philosophical Transactions of the Royal Society B: Biological Sciences, 353(1377), 1915–27. doi: 10.1098/rstb.1998.0344
- Rao, S. M., Leo, G. J., Bernardin, L., & Unverzagt, F. (1991). Cognitive dysfunction in multiple sclerosis.: I. Frequency, patterns, and prediction. Neurology, 41(5), 685–91. doi: 10.1212/wnl.41.5.685
- Reio, T., Czarnolewski, M., & Eliot, J. (2004). Handedness and spatial ability: Differential patterns of relationships. Laterality: Asymmetries of Body, Brain and Cognition, 9(3), 339–58. doi: 10.1080/13576500342000220
- Rourke, B. P., & Conway, J. A. (1997). Disabilities of arithmetic and mathematical reasoning. Journal of Learning Disabilities, 30(1), 34–46. doi: 10.1177/002221949703000103
- Sawcer, S., Franklin, R. J. M., & Ban, M. (2014). Multiple sclerosis genetics. The Lancet Neurology, 13(7), 700–9. doi: 10.1016/s1474-4422(14)70041-9
- Schoonheim, M. M., Hulst, H. E., Brandt, R. B., Strik, M., Wink, A. M., Uitdehaag, B. M. J., et al. (2015). Thalamus structure and function determine severity of cognitive impairment in multiple sclerosis. Neurology, 84(8), 776–83. doi: 10.1212/wnl.0000000000001285
- Spring, A., & Deutsch, G. (1981). Left brain right brain. San Francisco: WH Freeman.
- Thornton, A. E., & Raz, N. (1997). Memory impairment in multiple sclerosis: A quantitative review. Neuropsychology, 11(3), 357–66. doi: 10.1037/0894-4105.11.3.357
- Overell, J. R. (2011). Peripheral neuropathy: pattern recognition for the pragmatist. Practical Neurology, 11(2), 62–70. doi:10.1136/jnnp.2011.241612
- Warren, J. (2010).What should you tell patients with MS about their risk of developing dementia. ACNR, 10(3):20-21.
- Welsh, M. C., & Pennington, B. F. (1988). Assessing frontal lobe functioning in children: Views from developmental psychology. Developmental Neuropsychology, 4(3), 199–230. doi: 10.1080/87565648809540405
- Whelan-Goodinson, R., Ponsford, J., Johnston, L., & Grant, F. (2009). Psychiatric disorders following traumatic brain injury. Journal of Head Trauma Rehabilitation, 24(5), 324–32. doi: 10.1097/htr.0b013e3181a712aa
- Yektamaram, A., Nazmdeh, K. (2009). [During rehabilitation in multiple sclerosis patients (Persian)]. 1st Ed. Tehran: Welfare Organization Pub.
Type of Study: Original Research Article |
Subject:
Cognitive behavioral
Received: 2017/06/30 | Accepted: 2017/11/15 | Published: 2018/04/1
Received: 2017/06/30 | Accepted: 2017/11/15 | Published: 2018/04/1
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |